Tel: +86-17507082182
Email: sales@juvotron.com
WhatsAPP: +86-17507082182

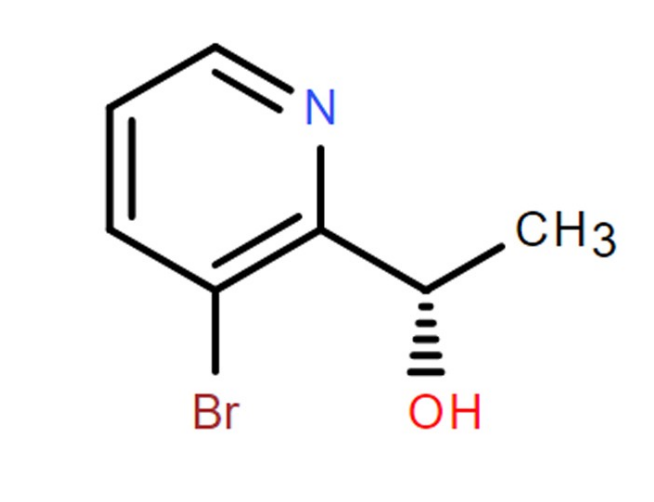

| Cas No. | 317845-81-1 |
| Molecular Formula | C7H8BrNO |
| Purity. | ≥99% |
| Molecular Weight | 202.05 g/mol |
| Chemical Name | (S)-1-(3-Bromopyridin-2-yl)ethan-1-ol |
| Synonyms |
317845-81-1;(1S)-1-(3-bromo-2-pyridyl)ethanol; (S)-1-(3-Bromopyridin-2-yl)ethan-1-ol;(S)-1-(3-Bromopyridin-2-yl)ethanol;(1S)-1-(3-bromopyridin-2-yl)ethanol;(1S)-1-(3-bromopyridin-2-yl)ethan-1-ol; (S)-1-(3-Bromo-2-pyridyl)ethanol |
| CAS No. | 317845-81-1 |
| Molecular Formula | C7H8BrNO |
| Molecular Weight | 202.05 g/mol |
| Appearance | White Powder |
| Storage and Shipping Conditions | |
| Industries | Pharmaceutical intermediates |
| Applications |
1. Pharmaceutical Field (Core Applications)
a. Pharmaceutical Intermediate: Used in the synthesis of pyridine-based targeted drugs, including antitumor agents (e.g., EGFR inhibitors), antibacterial drugs, and antiviral drugs. Its chiral structure and pyridine ring enhance the specificity of drug-target binding.
b. Drug Molecular Modification: Through derivatization reactions involving hydroxyl and bromine groups, it optimizes drug solubility, lipophilicity, and metabolic stability, thereby improving efficacy and reducing side effects.
c. Peptide and Small-Molecule Drug Construction: Serves as a chiral building block in the synthesis of heterocyclic peptide mimetics and alkaloid drugs, enriching the structural diversity of drug molecules.
2. Fine Chemical Industry
a. Chiral Synthesis Reagent: Functions as a chiral source in asymmetric synthesis for producing optically pure intermediates for dyes and fragrances. Its chiral hydroxyl group enables directional introduction into target molecules.
b. Organic Functional Material Precursor: Utilizes the coordination of pyridine rings with metal ions to prepare chiral catalysts and optically active polymer materials, meeting the needs for specialized functional materials. |